This text is adapted from and updates the introduction by G. Tran et D. Sauvant of the book Tables de composition et de valeur nutritive des matières premières destinées aux animaux d'élevage (Sauvant et al., 2004).
Feeds
The feeds presented in these tables are ingredients (also called feedstuffs, feedingstuffs, feed materials, or straights in English) used in compound feeds. Most of these feeds are common products, but some less conventional feeds, such as new feeds (insects...) as well as feeds used in tropical regions, have been included. The tables do no include forages and pastures. The feed names are those used in the feed trade. However, because feeds can be highly variable due to differences in genetics, cultivation, harvest and post-harvest conditions, and processing, more restrictive names based on the composition (typically protein, crude fibre and fat) have been created to provide profiles better suited to formulation.
Chemical data
Sources and methods
The chemical data used to calculate the values shown in the tables are obtained from analyses performed in private and public laboratories specialised in feed analysis (mostly in France and Western Europe). Analytical methods are those recommended by international and national standards organisations (such as ISO and AFNOR) and by other institutions (such as the European Commission and AOAC). In some cases, data are obtained by methods similar to or derived from standard ones, and by methods recommended by certain authors (such as those developed by Van Soest). Whenever possible, chemical compositions are established from data that are both recent (obtained in the past ten years) and available in sufficient numbers to allow critical evaluation. When such data are not available, data from the scientific literature are used instead. This is notably the case for vitamins and for part of the trace elements and fatty acids. The table below summarizes the main methods used to measure the chemical characteristics published in the tables.
| Characteristics | Methods |
| Moisture | Methods based on dessication, such as AFNOR NF V18-109 for usual feed materials. |
| Crude protein | Methods based on mineralization of nitrogen, such as the Kjeldahl method (i.e. AFNOR NF V18-100, 1977) or the Dumas one (AFNOR NF V18-120, 1997) for the more recent data. The crude protein content is obtained by multiplying total nitrogen by 6.25. |
| Crude fibre | “Weende” methods, based on acid hydrolysis followed by alkaline hydrolysis, such as AFNOR NF V03-40 (1993). Crude fibre is a default estimate of the cell wall content, which is actually 2 to 4 times higher. The crude fibre residue includes variable proportions of different cell wall constituents, such as lignin. |
| Crude fat | Methods based on the extraction of lipid substances by a solvent such as diethyl ether, hence the usual appellation of "ether extract". In France, the reference method for most feed materials is AFNOR NF V18-117 (1997). For animal products/byproducts, gluten, potato pulp, distillery and brewery byproducts, yeast, dairy products and bakery/biscuits byproducts, crude fat is extracted after HCl hydrolysis. |
| Ash | Methods based on incineration, such as AFNOR NF V18-101. |
| Insoluble HCl fraction | Residue after incineration and HCl treatment, e.g. AFNOR NF V18-102. |
| Cell wall components by Van Soest | Methods derived from the sequential method described by Van Soest (AFNOR NF V18-122, 1997):
|
| Water insoluble cell walls | Cell walls obtained by the AFNOR V18-11 method, including treatments with an alpha-amylase and a protease, followed by the removal of lipids. |
| Starch | Ewers polarimetric method, e.g. 3rd EC directive 72/199 modified 27/11/1980. Starch data obtained by enzymatic methods were not considered in the tables. It must be noted that this method may return values even when actual starch is absent or present in very limited amounts. |
| Total sugars | Most of the total sugars presented in the tables have been obtained by the Luff-Schoorl method (ethanol extraction). However, due to the lack of sugar values, enzymatic methods have also been taken into consideration. |
| Fatty acids | Methods based on the use of chloroform/methanol, methylation and extraction of methyl esters followed by chromatography. The fatty acids to crude fat conversion coefficients result from a compilation of literature data. |
| Amino acids | Acid hydrolysis (HCl 6N) followed by chromatography. There are many methods with variants in duration (24-48 hours) and temperature (110-145°C). Methionine and cystine are obtained after performic acid oxidation and tryptophan after alkaline hydrolysis. |
|
Minerals and trace elements |
Spectroscopic methods adapted for each mineral, such as AFNOR V18-108 for calcium and AFNOR NF V18-106 for phosphorus. Some values (particularly iodine) were derived from previous feed and food composition tables. |
| Phytate phosphorus | Organic plant phosphorus bound in phytic acid. Phytate phosphorus is calculated as 28.2% of phytic acid. Different methods can be used to measure phytic acid (precipitation of an iron composite or HPLC). |
| Phytase activity | One unit of phytase activity corresponds to the liberation of one µmol per minute of inorganic phosphorus from a solution of sodium phytate, measured at a fixed temperature and pH. |
| Vitamins | The data for vitamins come mainly from tables. The analytical methods were rarely indicated in the sources.
|
| Real applied viscosity | Real applied viscosity (Carré et al., 1994) is measured by viscosimetry of an aqueous extract. While potential applied viscosity is obtained after extraction with 80% ethanol, this treatment is not used for real applied viscosity in order to keep the endogenous enzymes active. Real applied viscosity values are not additive. Viscosity is expressed as:
|
| Dietary cation-anion difference (DCAD) and electrolyte balance (EB) | DCAD (for ruminants) and EB (for monogastrics) characterise the “acidifying” or “alkalising” potential of a feed or diet. This calculation integrates the ions with the greatest influence on the acid-base equilibrium: potassium and sodium are “alkalising”, and chlorine and sulphur are “acidifying”. Sulphur is not taken into account in the calculation of EB. DCAD and EB are expressed in mEq/kg of dry matter. Na, K, Cl and S are expressed in g/kg of dry matter.
|
Gross energy is measured by calorimetry or predicted from the chemical composition. In the tables, gross energy values are obtained using the following equation, which resulted from a statistical analysis carried out on more than 2000 gross energy values (Tran and Sauvant, 2000, unpublished):
GE = 17.3 + 0.0617 CP + 0.2193 EE + 0.0387 CF – 0.1867 Ash + Δ
GE: gross energy in MJ/kg of dry matter; CP, EE, CF and Ash represent crude protein, ether extract, crude fibre and minerals respectively, expressed in % dry matter. Δ: correction coefficient specifc to a feed group.
Δ values are presented in the following table:
| Feed group | Δ |
| Corn gluten meal | 1.29 |
| Blood meal | 1.12 |
| Alfalfa protein concentrate | 1.04 |
| Wheat distillery by-product, wheat gluten feed, maize bran, rice bran | 0.58 |
| Full fat rapeseed, full fat linseed, full-fat cottonseed, cottonseed meal | 0.49 |
| Oats, wheat milling by-products, corn gluten feed and other maize starch by-products, maize feed flour, sorghum | 0.31 |
| Dehydrated grass, straw | 0.19 |
| Barley | 0.15 |
| Barley rootlets, meat and bone meal | -0.18 |
| Linseed meal, palm kernel meal, full fat soybean, soybean meal, sunflower meal, sunflower seed | -0.19 |
| Cassava | -0.23 |
| Faba bean, lupin, pea | -0.36 |
| Sugar beet pulp, molasses, vinasse, potato pulp | -0.43 |
| Whey | -0.74 |
| Soybean hulls | -0.97 |
| Other feed materials except starch and brewer’s grains | 0.00 |
For maize starch and brewers grains, the equation proposed by Noblet (personal communication, 2002) was used:
GE = 0.2299 CP + 0.3893 EE + 0.1740 Starch + 0.1655 Sugars + 0.1884 NDF + 0.1773 Residue
Abbreviations and units are the same as in the previous equation. The Residue is the difference between organic matter and the sum of the other constituents in the equation.
Consistency
Some analytical parameters are more frequently measured than others, and, as a result, the number of observations available for calculations differs widely between parameters. For instance, crude protein values are much more common than amino acids values ; likewise, crude fibre values are much more common that Van Soest fibre values. When table values are calculated by averaging data sets of different sizes, this may result in inconsistent profiles: because of a lower number of raw values, the average ADF, for instance, will not correspond to the average crude fibre, which will result in turn in inconsistent energy values if the latter are calculated from equations based on ADF. In order to prevent these inconsistencies, thousands of regression equations between chemical parameters, most of them specific to a feed or group of feeds, have been established or collected from the scientific literature, and used to predict, for instance, fibre values and amino acid values consistent with the crude protein content.
Nutritional data
The nutritional data (energy, protein and mineral bioavailability values) are calculated according to state-of-the-art feed evaluation systems. These systems use equations specific to each animal species, which are described in the corresponding sections:
General process
The nutritional values are calculated to be consistent with the chemical composition, using equations derived from the statistical analysis of large data sets including in vivo and chemical data collected from INRA or from the scientific literature. The perimeter of these equations can be a single feed or a group of feeds. The input of these equations are the chemical data established previously. Many of these equations use cell wall constituents (crude fibre, NDF, ADF or lignin) as predictors, but other parameters are taken into account depending on the species, the feeds and the characteristics to be predicted. When relevant equations are not available, average values were used instead. When neither equations nor original data were available, nutritional data were derived from previous tables.
Energy values
Gross energy is not fully available to the animal: part of it is lost in faeces, urine, methane and heat increment. These losses can be estimated to provide energy values that represent the amount of energy actually used by the animal.
- Digestible energy (DE) is the energy available after deduction of faecal losses.
- Metabolizable energy (ME) is the energy available after deduction of faecal energy losses, urine energy losses and methane energy losses.
- Net energy (NE) (only for pigs, ruminants and horses) is the energy available after deduction of faecal energy losses, urine energy losses, methane energy losses and heat increment.
The figure below illustrates the different types of feed energy.
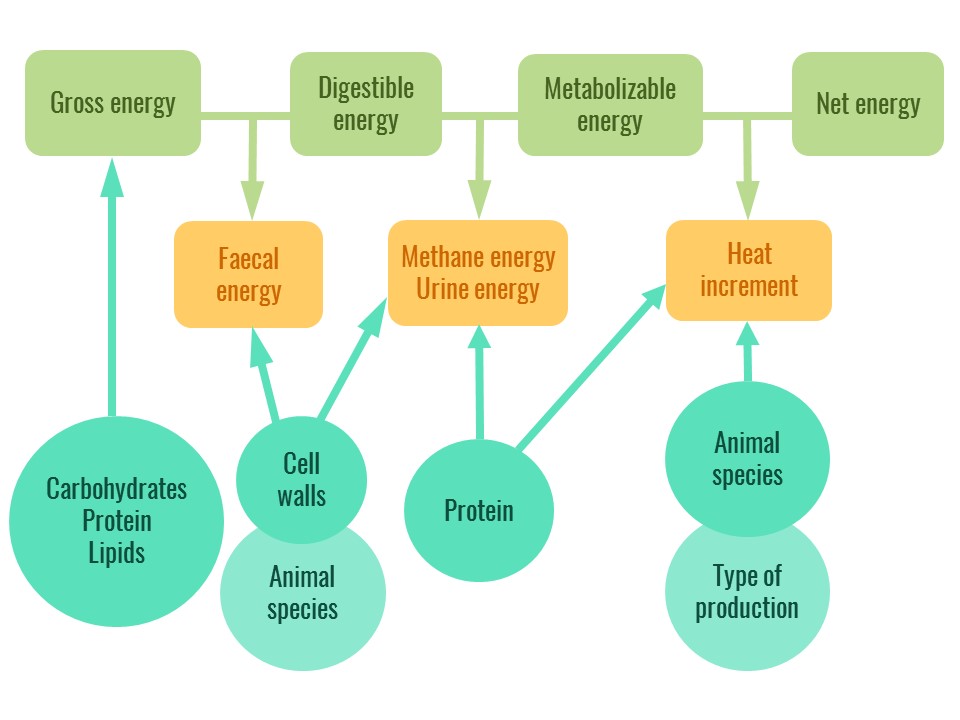
Figure 1. The different types of feed energy
The digestibility of energy (or of organic matter) can often predicted from the chemical composition, and particularly from the cell wall content ("fibre"), which is usually negatively correlated to it. These relationships depend on the animal species, as shown in the figure below.
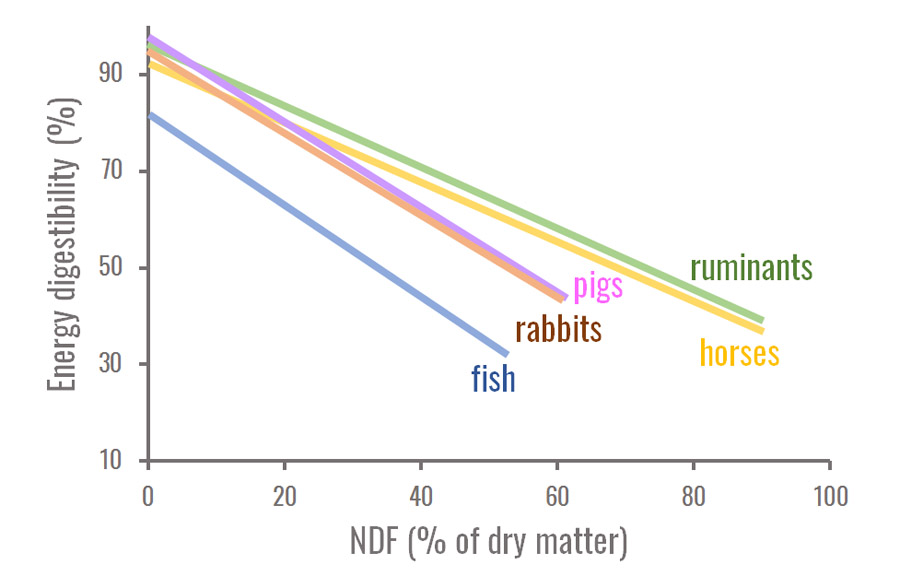
Figure 2. Comparison of the influence of cell wall content on energy digestibility for several species
DE is calculated as GE x energy digestibility, and ME and NE values are subsequently calculated using equations specific to each animal species that take into account the composition of the feed, the type of production and the physiological status of the animal (growing vs adult animal, milk vs meat production). The calculation processes are described for each species in the relevant sections.
- Ruminants: NE is provided as Forage Units (UFL for milk production, UFV for meat production) according to the new INRA Systali system (2016) and to the former INRA system (2007). Values are also provided in energy units (kcal/kg and MJ/kg).
- Pigs: DE, ME and ME are provided for growing and adult pigs.
- Poultry: only ME is provided, as energy losses in faeces and urine cannot easily be separated in poultry.
- Rabbits: DE and ME are provided. The tables used the digestibility data (with some modifications) and the concept of metabolisable energy published by Perez et al. (1998).
- Horses: NE is provided as Forage Unit (UFC).
- Salmonids: DE. The fish values were derived from the same sources used to establish the tables in Nutrition et Alimentation des poissons et crustacés (Guillaume et al., 1999).
Nitrogen values
Due to the differences between species concerning the digestive and metabolic use of ingested nitrogen, more or less elaborate estimations of nitrogen value are provided for each animal species, described in the corresponding sections.
- Ruminants:
- Nitrogen digestibility, intestinal nitrogen digestibility
- PDIA, PDI and Ruminal Protein Balance calculated according to the new INRA Systali system (2016)
- PDIA, PDIE and PDIN calculated according to the former INRA system (2007)
- Effective nitrogen degradability
- Levels of intestinal digestible essential amino acids are provided according to both systems
- Pigs: nitrogen digestibility and standardised ileal digestible amino acids (and amino acid digestibility), corrected for measured endogenous nitrogen losses, considered to be constant ("standardised"). These values were derived from the AmiPig tables published in 2000 by INRA, AFZ, Ajinomoto Eurolysine, Arvalis and Adisseo.
- Poultry: true faecal digestible amino acids, corrected amino acid for measured endogenous nitrogen losses.
- Rabbits: nitrogen digestibility.
- Horses: Digestible Crude Protein (MADC).
- Salmonids: nitrogen digestibility
Mineral values
Phosphorus
The majority of phosphorus contained in plant storage organs is in a form bound to phytic acid (myo-inositol hexaphosphoric acid). The release of this phosphorus requires enzymatic hydrolysis by a phytase. Phytate phosphorus is not available for monogastric animals because they do not possess sufficient intestinal phytase activity to hydrolyse the phosphate groups of this molecule. Therefore, a large part of the phytate phosphorus is not used and can be found in the excreta. However, it is possible to improve its availability by adding exogenous phytases to the feed. In addition, plant phytases contained in some feed materials, such as wheat, can hydrolyse the phytate phosphorus in the other diet ingredients (Nys et al., 1997). This problem is less important for ruminants since the microorganisms in the rumen produce phytases.
The tables present three types of nutritive values for phosphorus:
- Ruminants: absorbed phosphorus
- Pigs: apparent faecal phosphorus digestibility for the pigs (with in some cases different values depending on the denatured or non-denatured state of the endogenous phytase)
- Poultry: phosphorus availability for chickens
- Salmonids: apparent faecal phosphorus
Sources of minerals
The tables present the relative biological value (RBV) of the major sources of minerals (mineral products and organo-metallic complexes) used in Europe for pigs, poultry and ruminants. This review was performed by a group of experts commissioned by the EMFEMA using all the available literature data.
Environmental impact data
The environmental impact data have been produced through ECO-ALIM, a French project led by IFIP and funded by ADEME and the French ministry of agriculture. IFIP, Arvalis, Terres Inovia and INRA collaborated with the feed industry (SNIA, Coop de France Animal nutrition, AFCA-CIAL and feed manufacturers) to produce environmental data suitable for inclusion in least-cost feed formulation with a goal to reduce the environmental impact of livestock feeds and of the resulting animal products (meat, milk, eggs).
The environmental impact values of feeds were assessed by Life Cycle Analysis and are expressed per kilogram of feed. 6 parameters are included:
- Phosphorus consumption
- Cumulative energy demand (non-renewable: fossil+nuclear)
- Climate change
- Acidification
- Eutrophication
- Land competition
The characterization method CML-IA (Guinée et al. 2002) was chosen for the calculation of eutrophication and land use and ILCD method for Climate Change (CC), acidification (AC) and cumulative energy demand (CED).
References
- Blok M. C., Dekker, R. A., 2017. Table 'Standardized ileal digestibility of amino acids in feedstuffs for poultry'. CVB Documentation report nr. 61, November 2017 DOI: 10.18174/426333
- Carré B., Gomez J., Melcion J. P., Giboulot B., 1994. La viscosité des extraits aqueux des aliments destinés aux oiseaux d'élevage : méthodologie de mesure, effets des traitements technologiques, utilisation pour la prédiction des consommations et excrétions d'eau chez le dindonneau de chair de cinq semaines. Colloque annuel Valicentre, 13 déc. 1994, Chambray-les-Tours, CRITT Valicentre, Orléans, 66-77.
- EMFEMA, 2002. Bioavailability of major and trace minerals. EMFEMA, Bruxelles.
- INRA, 2010. Alimentation des bovins, ovins et caprins. Besoins des animaux - Valeurs des aliments. Editions Quae, Versailles, 315 p.
- INRA, 2018. INRA feeding system for ruminants. Wageningen Academic Publishers, Wageningen, the Netherlands, 640 pp. DOI: 10.3920/978-90-8686-292-4
- Perez J.M., Maertens L., Villamide M.J., de Blas C., 1998. Tables de composition et de valeur nutritive des aliments destinés au lapin : Conclusions d'un groupe de travail européen. In: Recherche cunicole (p. 141-146). Presented at 7. Journées de la recherche cunicole, Lyon, FRA (1998-05-13 - 1998-05-14). Paris, FRA : ITAVI - Institut Technique de l'Aviculture.
- Sauvant D., Perez J.M., Tran G., Association Française de Zootechnie, 2004). Tables de composition et de valeur nutritive des matières premières destinées aux animaux d'élevage (Porcs, volailles, ovins, caprins, lapins, chevaux, poissons). Paris, FRA : INRA Editions, 301 p.